
Are You Affected by the Clonazepam Recall?
The FDA Food and Drug Administration has issued a recall for 16 lots of Clonazepam manufactured by Endo, Inc., following a critical labeling error. The product safety recall is hoping to prevent medication errors. According to the safety alert, the strength listed on the product cartons (0.125 mg) did not match the actual strength of...
CONTINUE READING
Has Exactech Escaped Liability by Filing Bankruptcy?
Exactech is a global medical device company that creates orthopedic implant devices and surgical instrumentation for joint replacement surgeries. In June 2021, Exactech issued recalls of its knee, hip, and ankle implant systems due to defective packaging that caused the polyethylene inserts to oxidize prematurely. This failure led to rapid device deterioration and early implant...
CONTINUE READING
The Yost Legal Group Is Accepting Lawsuits Against Defective Drug Tepezza Do You Have a Case?
Teprotumumab (brand name Tepezza) is a United States Food and Drug Administration-approved medication used to treat the effects of thyroid eye disease (TED, also known as Graves’ disease). Historically, FDA approval is a long, winding, and costly process for pharmaceutical companies. New-drug development costs have skyrocketed in recent years to an average of more than...
CONTINUE READING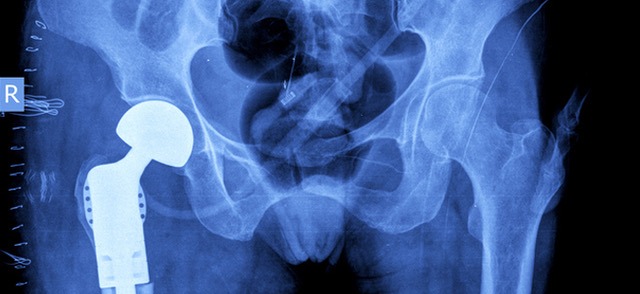
Missouri Woman Awarded $20 Million in Missouri Metal-on-Metal Hip Case
Mary Bayes and her husband Philip Bayes received their holiday presents early this year. A St. Louis federal jury awarded the couple $20 million and $1 million respectively for the injuries Mrs. Bayes suffered as a result of her defective Biomet M2a Magnum metal-on-metal hip replacement. Although the second trial, which ended recently, resulted in...
CONTINUE READING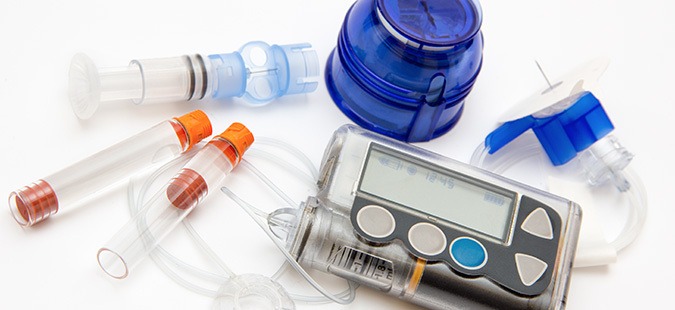
Medtronic Recalls Mini Med and Pro Infusion Insulin Pumps due to Life-Threatening Injuries
The MiniMed 600 series Insulin Pump System (630G and 670G) and its Pro Infusion Sets, in particular, is an example of this device used for diabetes management. Manufactured and distributed by Medtronic, the product appeared to be an effective therapy for diabetic patients. However, in response to numerous adverse reports, Medtronic voluntarily provided its customers...
CONTINUE READING
The Truth Behind Dangerous Medical Devices
Recently, attorney Tom Yost sat down with Elsa, from WMAR’s Midday Maryland to discuss the potential dangers of medical devices, specifically Essure birth control and metal on metal hip implants. He talks about the way in which the FDA approves these devices and allows them to be sold. As well as what the next steps...
CONTINUE READING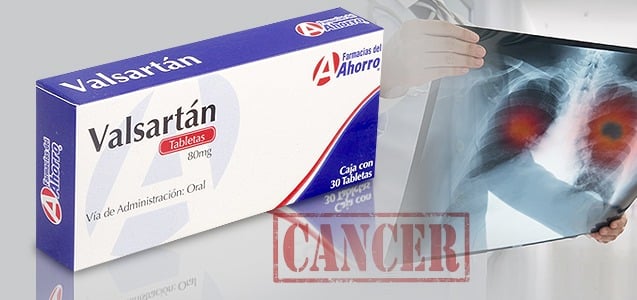
Dangerous carcinogens discovered in heart medication Valsartan
In July of 2018, concerns about cancer prompted the FDA (Food and Drug Administration) to issue a voluntary recall for the blood pressure and heart medication Valsartan. Doctors prescribe Valsartan for patients to treat their high blood pressure and congestive heart failure. Pharmacies are sending letters to patients who have been taking Valsartan, advising them...
CONTINUE READING
The Top 5 Most Significant Product Recalls of 2016
When making an important purchase, such as a mobile device or a vehicle, there are many factors that help in your decision making process. It is important to consider the potential safety risks associated with any product that you purchase. As consumers, we put our faith in manufacturers to deliver safe products and we trust...
CONTINUE READING
FDA Issues Safety Warning About Medicinal Clay Product Due to Elevated Levels of Lead
The Food and Drug Administration (FDA) is warning consumers to avoid “Bentonite Me Baby” by Alikay Naturals due to a risk of lead poisoning. “Bentonite Me Baby” is marketed as medicinal clay that can be ingested or applied topically for the treatment of several conditions. However, safety testing of this product revealed that that “Bentonite...
CONTINUE READING
Center for Disease Control Calls for More Research About the Safety of Using Antidepressant Medications During Pregnancy
In the CDC’s most recent Morbidity and Mortality Weekly Report, the agency calls for greater research on antidepressant drug use during pregnancy. The CDC’s research in the past has indicated that there may be a link between the use of SSRI antidepressant medications during pregnancy and an increased risk of serious birth defects in children....
CONTINUE READING