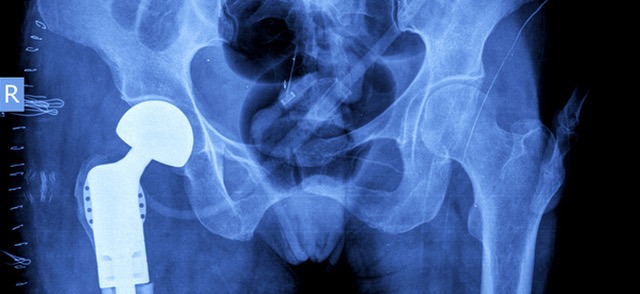
Defective Hip Implants Are Leading to Lawsuits
Do You Know What Brand of Hip You Have? Hips are in the spotlight right now on some of the biggest stages around the world. Andy Murray, Sybil Shapiro, and Al Roker have all made successful comebacks in their respective fields after hip resurfaces and replacements. Unfortunately, for many other people, hip replacements have only...
CONTINUE READING
Trunnionosis—Once Again, Proof That Metal and Hips Don’t Mix
Thirty years ago, trunnionosis surfaced as a diagnosis to characterize corrosion and wear occurring in hip replacements at the head-neck interface that connects with the thigh bone (femur). Historically, orthopedic surgeons often found corrosion or wear associated with hip implant failures in the area of the hip bone (acetabulum), but not the femur. More recently,...
CONTINUE READING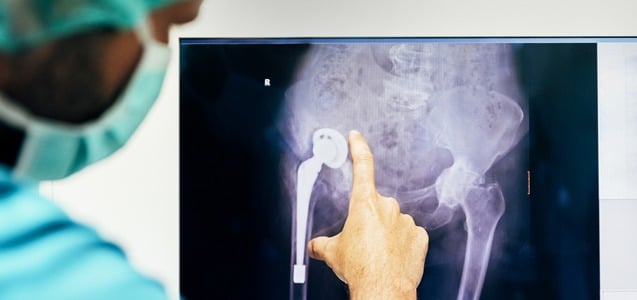
Metal on Metal Hip Replacements Linked to Serious Neurological Side Effects
A 2017 Health Watch study reported that elevated blood metal levels from metal-on-metal hip replacements are toxic to the brain. The findings of this research confirm an alarming and previously unknown risk of metal-on-metal hip replacements known as Arthroplasty Cobalt Encephalopathy (ACE). This condition includes a number of serious neurological side effects, including mood swings,...
CONTINUE READING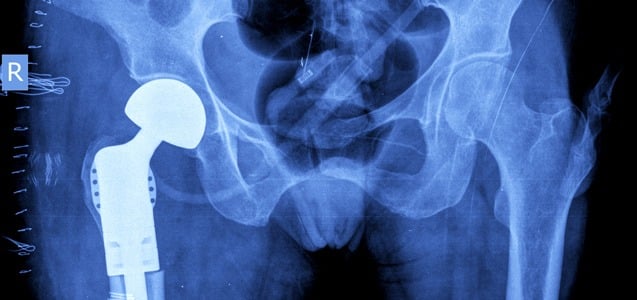
Stryker LFIT V40 Update
In August 2016, Stryker recalled its LFIT V40 femoral head hip implant based on higher than expected taper failure rates. A taper lock failure occurs when the femoral head of the hip implant comes loose from the stem of the hip implant. Many people implanted with this faulty device suffer from severe pain, inflammation, loss...
CONTINUE READING
Another Stryker Hip Recall: LFIT V40
The reputation of medical device manufacturer Stryker Corporation has been called into question in the past, after having to recall two of its metal hip replacement systems in 2012. More recently, the company once again faces criticism and outrage for its negligence in distributing yet another defectively designed hip replacement system. On August 29, 2016,...
CONTINUE READING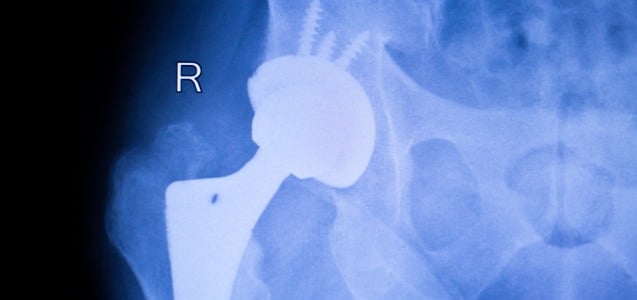
Stryker Issues Voluntary Recall Due To Defective Parts In Metal Hip Implant Devices
Stryker Corporation is one of the leading manufacturers of metal hip implant devices. In the past, defective Stryker metal hip implants have caused serious injuries and adverse reactions in thousands of patients. In some cases, patients have even required revision surgery in order to remove and replace the defective Stryker hip implant parts. Stryker recently...
CONTINUE READING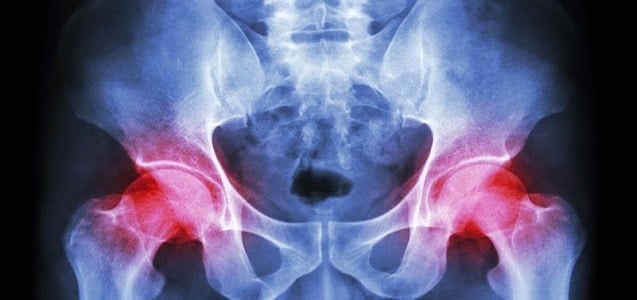
Judge Establishes June Deadline to File Claims Against Stryker Corp
Stryker Corp announced a major settlement agreement with thousands of former patients back in November of 2014, which could end up costing the company upwards of $1 Billion in pay-outs. A U.S District Judge in Atlanta, Georgia, announced on Monday, May 11 of 2015 that additional patients seeking damages against Stryker for their hip revision...
CONTINUE READING
Stryker Corp Faces Another Large Settlement in Fraud Case
Stryker Corporation has announced a major settlement of almost $1.5 billion dollars to be paid to thousands of patients injured by their metal-on-metal Rejuvenate and ABG II Model hip implants. The company now finds itself at the center of another controversy involving one of its subsidiaries, OtisMed, and another dangerously defective piece of medical equipment....
CONTINUE READINGStryker Rejuvenate and ABG II Modular Hip System Recall
Thousands of Americans receive hip replacements every year due to sports injuries, falls, aging and a host of other reasons. The goal of a hip replacement surgery is to return to your active lifestyle as quickly as possible. There are many different hip replacement medical devices on the market, from plastic implants to metal on...
CONTINUE READING