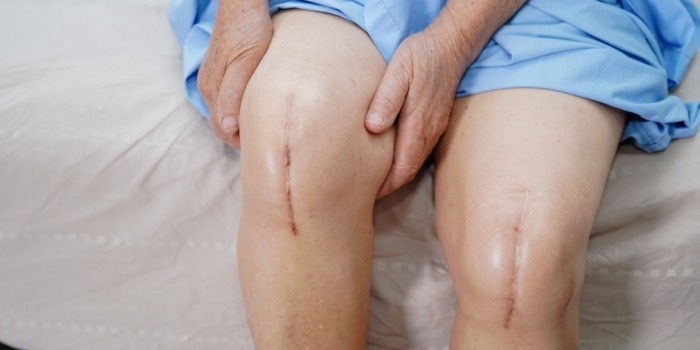
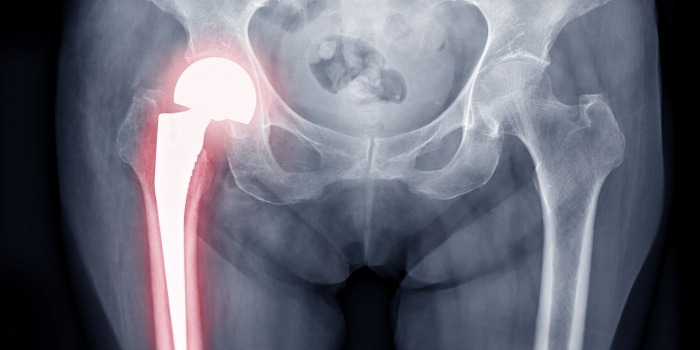
Exactech Hip Replacement Lawsuit Update 2023
Protect your rights by filing an Exactech Hip Replacement Lawsuit. Exactech is a global medical device company that creates orthopedic implant devices and surgical instrumentation for joint replacement surgeries. As a result of premature device failure, thousands of people require revision surgery or have developed serious health issues, such as Osteolysis.
If you or a loved one had any of the following surgeries, and the Exactech implant was used, you may qualify to file a claim against Exachtech.
– knee replacement surgery
– hip replacement surgery
– ankle replacement surgery
Did you have revision surgery for your hip replacement, knee replacement, or ankle replacement?
Did you receive a letter saying you require revision surgery?
Have you been diagnosed with Osteolysis after having hip, knee, or ankle replacement surgery?
Did you have hip replacement complications from the Exactech hip recall?
Protect yourself with the help of experienced Exactech lawyers
If you answered yes to any of the above, contact The Yost Legal Group at 800-967-8529 for a free, confidential consultation. We will answer your questions and inform you about the current Exachtech litigation. You may qualify to file a Hip Replacement Lawsuit.
Lawsuits have been filed against Exactech by victims seeking compensation for:
– past medical expenses
– future medical expenses
– pain and suffering
Exactech Hip Replacement Lawsuit Timeline
May 2023
Judge Donna Keim of Gainesville, FL, organized a special “science day” focused on Exactech implants. Two esteemed judges, Judge Nicholas Garaufis and Judge Marcia M. Henry from the Eastern District of New York, were invited to participate.
During this unique gathering, experts representing the plaintiff and defense sides of the Exactech lawsuit shared valuable insights on various aspects of the case.
Topics included the science behind hip and knee replacements, revisions, the materials used in the recalled Exactech implants, and the packaging materials. Additionally, other issues related to the recalled devices were also addressed.
Science Day was a significant opportunity for the judge to deepen her understanding of the recalled devices and raise pertinent questions. Moving forward, regular case management conferences will be held by the judges as the discovery phase progresses in the Exactech lawsuits. This ensures a thorough and practical examination of the matter at hand.
March 2023
The FDA warns patients and healthcare providers who have used Exactech joint replacement devices between 2004 and August 2021. They communicated about the recall of specific devices manufactured during this period.
The recalled Exactech joint replacement devices, including knees, ankles, and hips, were found to have been packaged in defective packaging bags. These bags lacked an essential oxygen barrier layer, which serves to protect the devices from oxidation – a chemical reaction that can deteriorate plastics over time.
The consequences of oxidation can be severe, resulting in accelerated device wear/failure, as well as component cracking or fracture. These issues may ultimately necessitate corrective revision surgery. It is important to note that some of the recalled devices are also associated with an increased risk of revision surgeries and bone loss due to excessive device wear/failure.
The FDA urged patients and healthcare providers to be aware of this recall and take the necessary steps to address potential risks or concerns. It was advised to contact the appropriate healthcare professionals to discuss further actions, such as device replacement or additional monitoring.
January 2023
Exactech pushes for official coordination and consolidation of federal and state court litigations related to the Exactech implant recalls.
December 2022
A rapid increase in lawsuits related to the Exactech recall has been filed into the Exactech MDL 3044.
October 2022
Due to the volume of cases filed, lawsuits involving the recalled knee and ankle products were consolidated into a Multi-District Litigation (MDL) in the Eastern District of New York. Similarly, many recalled hip and ankle products filed in Florida remain consolidated in the Florida state courts. Over a year later, over 850 lawsuits are pending in the MDL, and over 200 are pending in the Florida courts.
February 2022
To avoid paying patients what they truly deserve, Exactech chose early on to provide a settlement option upfront for its victims. Exactech retained the services of Broadspire, a third-party settlement administrator and risk management firm, to handle the claims reimbursement process.
Patients were encouraged to file a claim with Broadspire and obtain reimbursement for their out-of-pocket medical expenses only.
Essentially, Broadspire was hired by Exactech to lure victims in by appealing to people’s desire for quick money without the need for a lawyer. However, it is essential to remember that Broadspire is a risk management company whose sole purpose was to minimize the financial damage to Exactech.
The Broadspire settlement option only reimburses patients for medical expenses paid that insurance did not cover. This significantly reduces the amount of financial compensation Exactech will have to pay out.
Lost wages, related medical bills, and non-economic damages, such as pain and suffering, are not part of the calculus. A settlement through Broadspire will NOT compensate you for all of your damages.
Submitting a claim through Broadspire will impact your legal options against Exactech.
June 2021
Exactech issued recalls of its knee, hip, and ankle implant systems. They said defective packaging caused the polyethylene inserts to oxidize and deteriorate rapidly. This ultimately led to a premature failure of the devices.
At The Yost Legal Group, patients’ well-being is our top priority. We are here to provide support and guidance throughout this process. Please stay informed and take the necessary precautions to ensure your safety.
Get the compensation you deserve for a Defective Exactech Medical Device
If you or a loved one has an Exactech hip or knee implant and still need to file a claim against Exachtech, call one of our experienced Exactech lawyers for help. A product liability lawyer at The Yost Legal Group will fight to recover your maximum settlement.
Even if you have already received a settlement through Broadspire, we strongly advise you to speak with one of our knee replacement recall attorneys today.
Call The Yost Legal Group at 1-800-YOST-LAW (1-800-967-8529) or email us at info@yostlaw.com today for a free consultation. There is no fee or expense unless you recover.
If you had an Exactech knee replacement failure or revision hip replacement, we would file an injury claim for you. Our injury lawyers will review your medical records and analyze the premature wear of your medical device.
We will talk with you about your loss of “quality of life” because of your failed medical device and how it has impacted your life.
The Yost Legal Group has years of experience handling defective product cases. We will file an Exactech lawsuit and ensure your rights are fully protected. If you have a failed knee replacement or failed hip replacement, call us.
We will support your fight for justice and hold Exactech rightfully accountable for their defective products.
Why contact The Yost Legal Group personal injury lawyers
Our personal injury law attorneys have years of experience representing medical malpractice, product liability, and defective medical device claims. We are filing Exactech lawsuits against the manufacturer. The Exactech recall has affected thousands of patients.
An Exactech recall lawsuit will protect your rights to receive financial compensation to cover your pain and damages.
Our Exactech attorneys are here to explain the hip implant failure lawsuit and knee implant failure lawsuit. If you have had a hip revision surgery or knee revision surgery due to a defective Exactech implant, we are here to help.