A recent study from the British Medical Journal highlights a significant concern regarding Depo Provera birth control injection, linking its use to an increased risk of developing intracranial meningiomas or brain tumors.
The study shows that women who use the Depo contraceptive for more than a year have a 5.6 times higher risk of developing these tumors.
Pfizer’s Depo-Provera (Depo) was approved by the FDA in 1992 for the use of contraception. The progestogen hormone injection is administered every three months. Besides birth control, Depo is also used to treat conditions like endometriosis, ovarian cysts, and certain cancers such as uterine and kidney cancer.
If you have been diagnosed with brain tumors after using Depo Provera birth control, contact The Yost Legal Group. Our dangerous drug lawyers are filing medication lawsuits for women across the United States.
We will help you understand your rights and discuss the potential of filing a Depo Provera lawsuit against the drug manufacturer.
You may be entitled to financial compensation. The experienced product liability lawyers at The Yost Legal Group are here to help. Please call or text us at 1-410-659-6800 for a free consultation.
Important Facts About Meningiomas (Brain Tumors) and Their Impact
Forty percent (40%) of tumors found in the central nervous system are meningiomas. These tumors are often found on the dura (thin layer of tissue that covers the brain) at the base of the skull or near the top and outer curve of the brain.
Meningiomas typically occur outside the brain – pushing on it as opposed to arising within it. Even though meningiomas are mostly non-cancerous and slow-growing, they can have devastating neurological impacts, making surgical intervention and sometimes even radiation necessary.
Brain tumors are graded based on how aggressive they are. As such, there are three different types of meningiomas:
– Grade 1 tumor is the most benign, most slow-growing, and most commonly occurring
– Grade 2 tumor, or atypical meningioma, is more invasive – invading the brain and having a higher likelihood of growing back after surgery
– Grade 3 meningioma, or anaplastic or malignant meningioma, is very aggressive and has a much higher recurrence rate than an atypical (Grade 2) meningioma.
What Are Meningiomas and Symptoms of Brain Tumors
Meningiomas are slow-growing tumors that often develop without noticeable symptoms for years. When symptoms do appear, they progress gradually and can be quite subtle.
The symptoms vary based on the brain tumor’s location and may cause the following symptoms:
– Headaches
– changes in vision
– hearing loss
– memory issues
– seizures
– weakness
– paralysis.
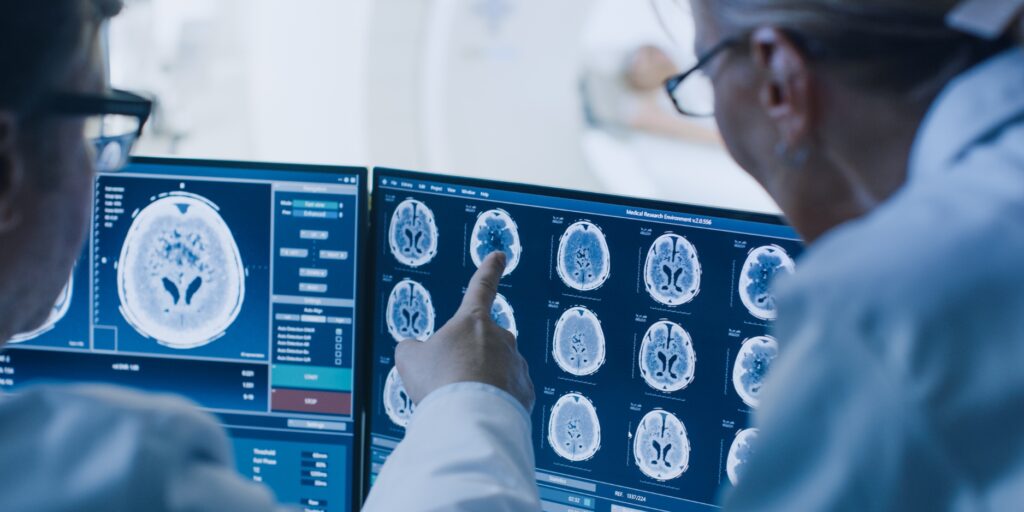
Meningiomas are most commonly detected through MRI scans. However, due to their subtle symptoms, they are often discovered incidentally during routine scans for other conditions or after head trauma.
As the majority of meningiomas are Grade 1 (benign and slow-growing), a more conservative approach to treatment is the usual standard of care. For example, a smaller, asymptomatic meningioma may be monitored with regular MRI scans rather than surgery.
However, surgery is usually the best treatment option if a tumor causes symptoms, is large, or shows growth during periodic monitoring.
Surgery vs. Radiation for Meningioma Treatment
For most patients, especially those with Grade 1 meningioma, successful surgery will remove the tumor completely.
For patients where surgery is not an optimal choice, targeted radiation therapy can be used to help stop the growth of the tumor.
While surgery is typically the end of treatment for benign meningiomas, patients should continue to have scans performed regularly to be sure that the tumor does not come back.
For more aggressive or malignant meningiomas (Grade 2 and Grade 3), those patients will likely require both surgery and radiation for treatment.
A Closer Look at Depo Provera’s Link to Brain Tumors
In recent years, birth control options have expanded significantly, offering women greater autonomy over their reproductive health. However, with these advancements come potential risks that must be carefully considered.
When the groundbreaking study was published in the British Medical Journal, it brought attention to the concerning link between the Depo shot and an increased risk of developing intracranial meningiomas.
This revelation has sparked discussions among healthcare professionals and patients about the safety of the long-term use of this birth control shot.
Intracranial meningiomas are generally non-cancerous tumors that arise from the meninges—the protective layers surrounding the brain and spinal cord—but can still cause significant health issues depending on their size and location.
These findings underscore the importance of informed decision-making when it comes to choosing contraception methods like Depo Provera birth control.
While its multifaceted applications make it an appealing option for many women, these latest findings necessitate revisiting its risk profile.
Depo-Provera and Brain Tumors: Understand Your Legal Rights
At The Yost Legal Group, we are dedicated to helping people affected by hidden risks linked to Depo-Provera. The side effects of the Depo shot can cause significant medical side effects.
If you or a loved one has been diagnosed with a brain tumor following prolonged use of this drug, explore your legal options. You may qualify to file a Depo Provera Cancer Lawsuit. Prescription drugs must have adequate warnings about their products.
Pfizer’s failure to warn women about the effects of Depo Provera has caused many to suffer significant harm. Our defective drug attorneys dedicate themselves to holding pharmaceutical companies accountable. We fight to prioritize patient safety over profits.
If you or a loved one has been diagnosed with a meningioma after taking Depo-Provera, you may have a claim.
Call or text us at 1-410-659-6800 for a free case evaluation. Seek the compensation you deserve by filing a lawsuit with an experienced product liability attorney.
Our defective product lawyers do not charge anything upfront. No charges or fees are due until after a recovery is made on your behalf. If there is no recovery, no fees or expenses are due.
Call our lawyers for negligence today to help you get the justice you deserve.
Birth Control Injection – Depo Injection Birth Control – Lawsuit Depo Provera – Depo Provera Birth Control
Depo Lawsuit – Birth Control Shot Lawsuit – Depo Birth Control Lawsuit