Attention ladies! We want to make sure you are well informed about a serious health risk involving Allergan breast implants. These defective implants may cause a rare and aggressive type of cancer called Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL).
This type of lymphoma is typically caused by a combination of textured implants and a compromised immune system. Allergan breast implants have been recalled due to the risk of ALCL Lymphoma.
Early BIA-ALCL symptoms include:
- Unexplained breast enlargement.
- Asymmetry.
- Mysterious skin rash.
- Fluid buildup.
- A lump in the armpit or breast.
Your well-being is our priority, so please take this information seriously and take action if you experience any of these symptoms. Stay vigilant and stay safe!
Legal Help for Those Impacted by the BIA-ALCL Risk
Textured implants are the implants of choice for both reconstructive and cosmetic breast implant recipients. They are much more realistic than smooth implants. Furthermore, Allergan is the leading manufacturer of textured implants.
They insisted that its products were safer than smooth implants. In a way, that is true. Textured implants are less likely to leak or rupture than smooth implants. However, the increased risk of developing BIA-ALCL is a poor trade-off in safety.
As of May 2023, over a thousand women have filed legal actions that are now pending in a New Jersey federal court. Allergan hasn’t made a settlement offer yet, but many think it will happen soon.
Victims of Allergan’s defective breast implants who partner with an experienced breast implant attorney are better equipped to navigate the settlement and disbursement processes.
If you or a loved one has been diagnosed with BIA-ALCL and have Allergan’s textured breast implants, you may have a case. Call The Yost Legal Group at 800-967-8529 for a free consultation about your rights and potential claim.
The Connection Between Allergan Textured Breast Implants and Cancer
Textured implants are designed with deeper and larger divots. This helps reduce the risk of leakage and ensures a more secure attachment to the chest. However, this type of implant can sometimes lead to the formation of scar tissue capsules.
While this soft capsule typically does not cause discomfort, it puts women at risk for BIA-ALCL without them knowing.
It’s important to note that lymphoma is not the only health concern associated with these implants. The cancer cells that can develop in the scar tissue have the potential to spread throughout the body and cause other types of cancer, as stated by the Food and Drug Administration.
Treatment usually means a combination of surgery to remove the implant and affected tissue, as well as radiation and chemotherapy. If doctors aggressively treat this aggressive disease, it is usually manageable. However, not all BIA-ALCL survivors can tolerate such treatments.
As mentioned, these implants are common in reconstructive surgery. If the survivor has already survived breast cancer or another serious illness, aggressive cancer treatments are impossible.
Furthermore, these treatments are expensive, usually over $10,000 per month. A group health insurance company might pay part of the cost. Only an experienced defective breast implant lawyer can obtain compensation for all the costs associated with treatment.
What breast implants and tissue expanders were affected in the Allergan recall?
In August 2022, the FDA received over 1,000 textured breast implant health complaints. Over 80% of these reports involved an Allergan product such as:
● Natrelle 410 Highly Cohesive Anatomically Shaped Silicone-Filled Breast Implants
● Natrelle Inspira Silicone-Filled Breast Implants
● Natrelle Saline Breast Implants
● Natrelle Silicone-Filled Breast Implants
● McGhan BioDimensional Silicone-Filled Biocell Textured Breast Implants
● McGhan Magna-Site Tissue Expander
● McGhan Style 134 Croissant Shaped Tissue Expander
● Natrelle 133 Plus Issue Expanders
● Natrelle 133 Tissue Expanders with and without suture tabs
● Style 133 Biospan Tissue Expanders
Important health warning regarding breast implants
We care deeply about your well-being and want to keep you informed about the latest developments in breast implant safety.
In 2011, the FDA identified a potential link between breast implants and a rare form of lymphoma called anaplastic large-cell lymphoma. However, at the time, the number of cases was too few to fully understand the risk.
In 2016, the World Health Organization classified this disease as a type of T-cell lymphoma that can develop after receiving breast implants. The following year, the FDA announced that textured implants were primarily associated with this condition, paving the way for a recall.
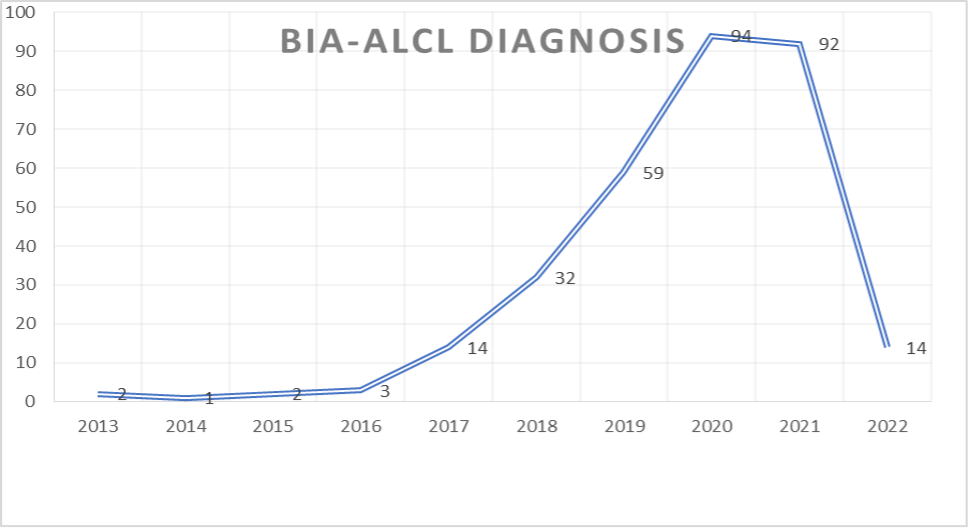
In March 2019, the FDA held a public advisory committee meeting to address the risks. Initially, a recall was not recommended, as product recalls are considered a last resort. However, in July 2019, new data revealed a concerning increase in cases. This undeniable evidence prompted the Allergan textured implant recall in July 2019.
In August 2022, the FDA received an alarming number of over 1,000 health complaints regarding textured breast implants. Shockingly, more than 80% of these reports were specifically linked to an Allergan product.
Your health and safety are our top priority at The Yost Legal Group. We want to ensure you have the information you need to make informed decisions. Please stay informed and consult with your healthcare provider if you have any concerns or questions regarding your breast implants.
How to file a legal action against Allergan for dangerous breast implants
Protecting customer safety should always be a top priority for companies. Unfortunately, Allergan missed multiple opportunities to inform customers about the heightened risk of developing BIA-ALCL after receiving their implants.
When a company values profits over people, it becomes crucial for a personal injury attorney to step up. The Yost Legal Group is now filing legal actions for negligence or failure to warn women nationwide against Allergan.
Call to speak with us about the Allergan implant recall. We’ll answer your questions about the Allergan lawsuit. Our breast cancer lawyers are here to protect your rights and to seek the maximum Allergan implants compensation for you.

Preparing for Your Free Case Evaluation Consultation
Below are some questions that will help our law firm determine if you have a case:
- Have you been diagnosed with Breast Implant-Associated Anaplastic Large-Cell Lymphoma?
o A current diagnosis is essential in a negligence claim. Note that BIA-ALCL is not the same as breast cancer. Instead, it is a type of non-Hodgkin lymphoma.
- When did you have your breast implant surgery?
o People filing lawsuits had their surgeries between 1998 and 2019. Contact your plastic surgeon who performed this procedure if you don’t remember the exact date of your surgery.
- What style of Allergan implants do you have?
o Most women do not pay much attention to the make and model. Not all Allergan breast implants have been known to cause BIA-ALCL. They would be textured implants.
- Have you had your implants removed?
o The FDA has not advised patients to remove their implants if they do not have symptoms of BIA-ALCL or another problem. However, you should hire a breast implant lawyer to protect your rights, especially if you have already had your implants removed.
Hire a Dedicated Defective Breast Implant Lawyer
Are you or a loved one suffering from BIA-ALCL due to Allergan’s textured breast implants?
The Yost Legal Group is here to provide compassionate assistance and fight for your rights. You may be eligible for substantial financial compensation. This will help ease the burden of medical bills, therapy expenses, and emotional distress.
Contact our experienced Allergan textured breast implant lawyers today for a free and confidential consultation. Call The Yost Legal Group at 1-800-967-8529.