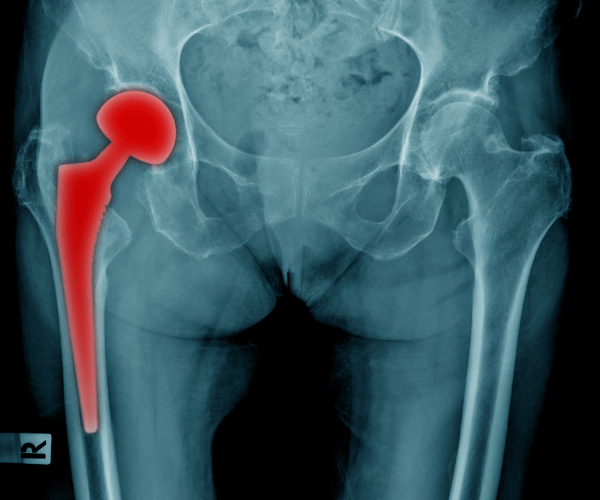
The U.S. Food and Drug Administration prompted medical-implant manufacturer Exactech to expand its voluntary hip implant recall to all models, not just some of them.
Exactech joint replacement devices are used to replace painful, arthritic joints due to osteoarthritis, osteonecrosis, rheumatoid arthritis, and loss of normal structure and function in adults.
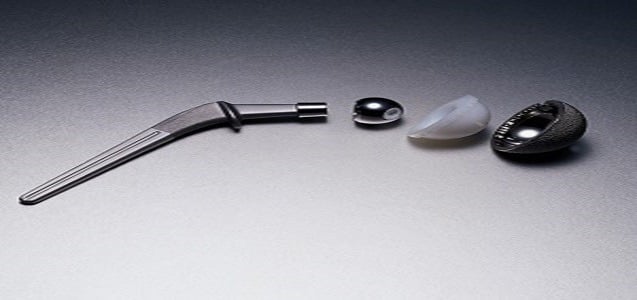
These devices are also used to improve previously failed joint replacement devices with adequate bone and soft tissue. All Exactech joint replacement devices contain a plastic component which should be in packaging that contains multiple oxygen-barrier layers as indicated in the package specification. The recalled devices were packaged in defective bags that were missing one of the oxygen barrier layers that protect devices from oxidation.
The FDA continues to work with Exactech to assess whether any of its other joint implants that contain polyethylene components packaged in defective bags may result in increased oxidation and similar risks.
Voluntary Hip Implant Recalls
A hip implant or other product recall is like closing the barn doors after the horses run off. When the FDA recalls a dangerous medical device, people have already been hurt or at least put at unnecessary risk for serious injury. Furthermore, a recall does not compensate these survivors for their economic losses (medical bills) and noneconomic losses (pain and suffering). Only an experienced defective-medical-implant lawyer can help victims get the justice the deserve.
Additionally, an FDA “recall” is not exactly what most people would think of a “recall.” By law, the Food and Drug Administration cannot unilaterally recall products, no matter how dangerous they are. In a few cases, FDA bureaucrats can limit marketing activities or unilaterally take another action that is not particularly effective. But in most cases, the FDA can only publicly pressure companies to voluntarily recall their own products.
Even then, a product recall is usually a sales modification. The company adds a warning to a dangerous product and keeps selling it. Warning labels, especially in defective hip implant cases, create several legal issues. More on that below.
Exactech Hip Implant Issues
Metal-on-metal hip implants have a long and unsafe history. Most of these defective hip implants have design or manufacturing defects. However, Exactech’s defective implants have packaging issues. Therefore, the product is defective before a doctor even implants it.
Once upon a time, hip implant durability was not an issue. Most hip implant recipients were semi-retired people who were not physically active. Then, in the late 1990s into the early 2000s, people in their fifties and even forties started getting replacements. To cash in on this uptick, manufacturers began taking shortcuts.
But we are getting a little ahead of ourselves. Some metal-on-metal hip implants were defective before they reached consumers. The hip is basically a cup-and-socket joint. When the all-metal parts grind together, the friction releases tiny metal fragments into the body and causes metallosis (metal poisoning).
Most manufacturers addressed this design defect by replacing some of the metal parts with ceramic parts. But the design defect was only part of the problem.
To cope with the increased demand, many hip implant manufacturers bought inexpensive parts from China and other overseas countries. These countries often have lax or nonexistent product safety laws. Therefore, many imported implant parts had high levels of mercury, cadmium, and other dangerous heavy metals.
The human body cannot process these toxic particles in unnatural amounts, so they accumulate in the body and cause a host of serious health problems.
Exactech hip implants, however, have a unique problem. The packaging does not properly prevent oxidation. A loaf of bread gets stale if you do not tie it up properly to keep the air out. Similarly, hip implants degrade if they are not tied up properly to keep the air out.
These defects lead to premature device failure. While routine, implants are still an invasive surgery, especially on older individuals. Once the device fails after implantation, a surgeon must fish the broken device out of the body and replace it with a new device—yet another invasive procedure. These multiple surgeries usually rob survivors of what little mobility they have left, assuming they are candidates for such risky procedures in the first place.
Your Legal Options
Survivors need and deserve financial compensation for device failure and other injuries. Additionally, the manufacturers who sold such products should accept responsibility for causing such problems in the first place. If a personal injury attorney files a legal action, that filing kills two birds with one stone. Defective implant survivors receive compensation. At the same time, companies face financial and reputational consequences.
Most attorneys file negligent failure to warn legal actions in these cases. Companies pride themselves on quality control measures. These companies are fully aware that the products they sell are defective. They just hope that if survivors file legal actions, they can beat those legal actions in court. These legal actions have basically four components:
- Duty: Exactech and other companies have a duty to identify device safety issues—such as improper oxidation protection—and a duty to warn customers about them. This warning must be proportionate to the risk.
- Breach: Exactech and other companies breach their duty of care when they do not warn customers or do not adequately warn them (e.g., placing a device failure warning on a long list of mild side effects). A personal injury lawyer typically partners with an industry expert who establishes the standard of care and testifies about the breach of care.
- Cause: In most states, survivors must prove, by a preponderance of the evidence (more likely than not), that the failure to warn substantially caused their injuries. The standard is slightly different in some states. Plaintiffs must also establish foreseeability (possibility) of injury.
- Damages: Device failure causes physical injuries. Device failure also causes emotional injuries. Compensation is available for both.
Assumption of the risk is one of the most common defenses in weak-warning cases. Companies like Exactech include serious side-effect warnings in the fine print. These warnings could reduce compensation in the case if the survivor saw the warning, could read the warning, and could understand what the warning meant.
In most states, jurors must divide injury responsibility on a percentage basis. For example, jurors might conclude, based on the evidence, that the company was 80% at fault for using defective packaging, and the survivor was 20% at fault for not heeding the warning.
Work with a Compassionate Defective Exactech Implant Lawyer
If you or a loved one had a defective Exactech hip implant removed after suffering harm, you may have a case, and The Yost Legal Group wants to help you seek justice. Injury victims are entitled to significant compensation for the harms they suffered. Call today: 1-800-967-8529.