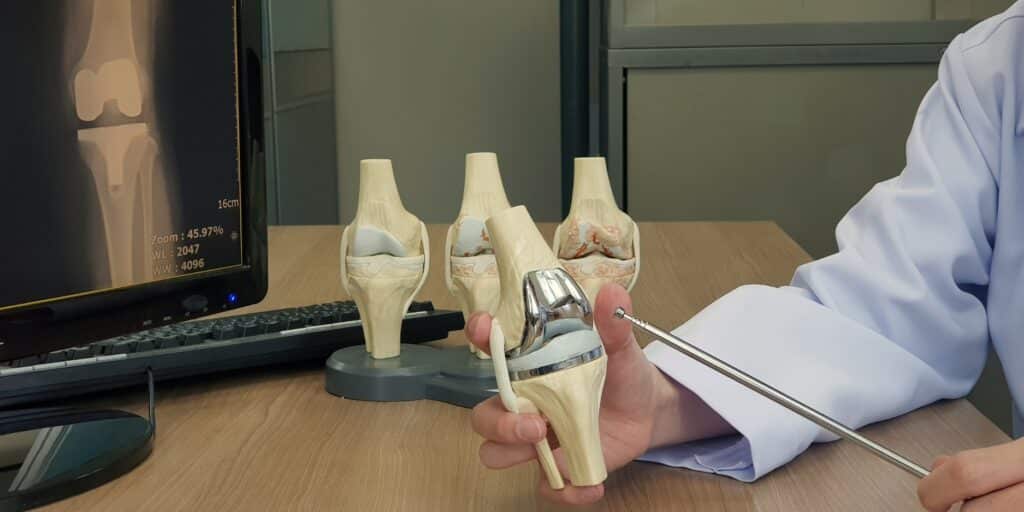
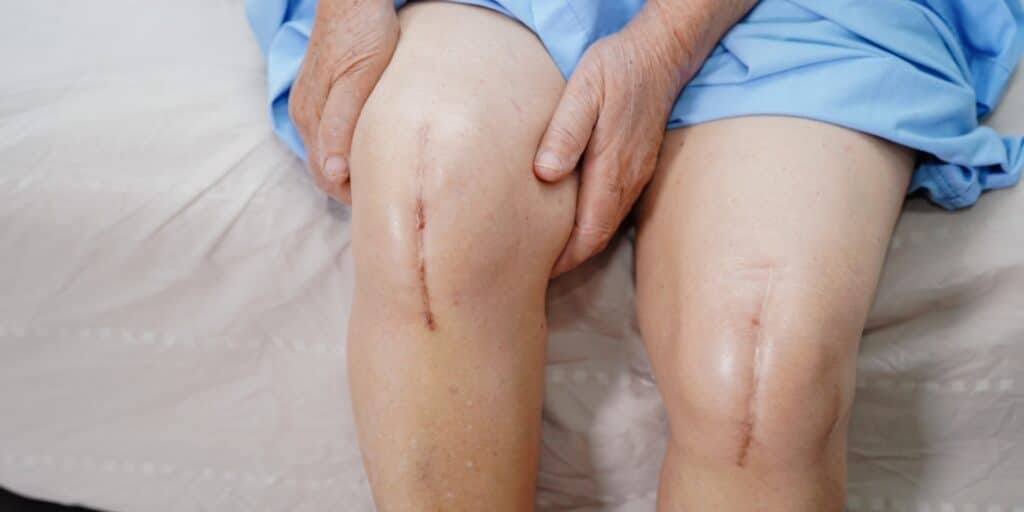
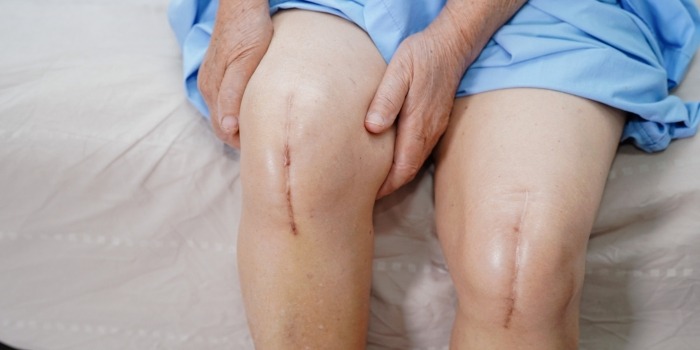
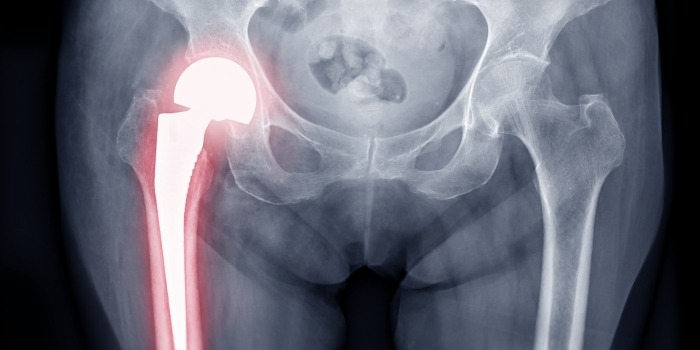
Exactech is a global medical device company that creates orthopedic implant devices and surgical instrumentation for joint replacement surgeries.
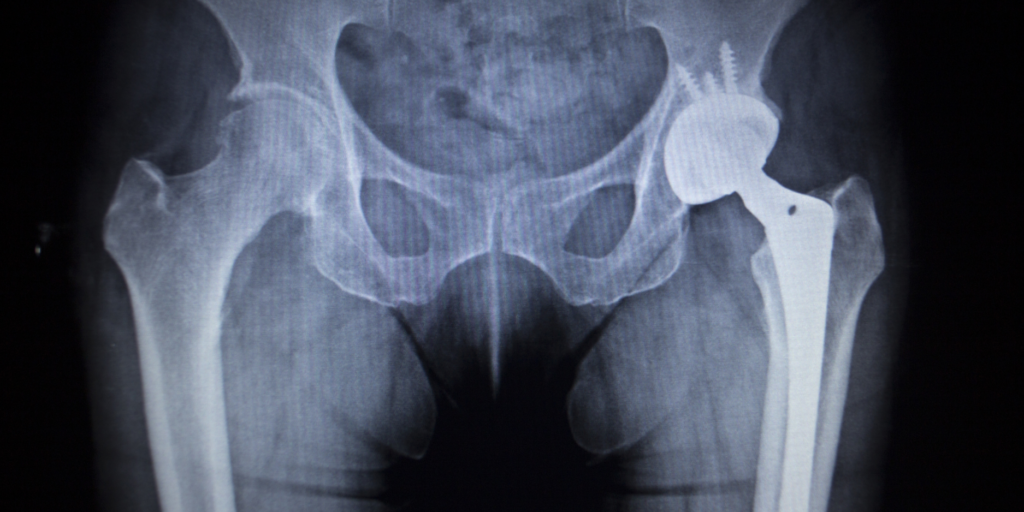
In June 2021, Exactech issued recalls of its knee, hip, and ankle implant systems due to defective packaging that caused the polyethylene inserts to oxidize prematurely. This failure led to rapid device deterioration and early implant failure.
As a result of this premature device failure, thousands of implantees required revision surgery or developed serious health issues, such as osteolysis.
Lawsuits have been filed against Exactech by patients seeking compensation for their past and future medical expenses and pain and suffering caused by the defective implants.
Due to the volume of cases filed, the lawsuits involving the recalled knee and ankle products were consolidated into a Multi-District Litigation (MDL) in October of 2022 in the Eastern District of New York.
Similarly, several cases of recalled hip and ankle products filed in Florida remain consolidated in the Florida state court.
Over 3000 lawsuits are collectively pending in the MDL and Florida state court.
Exactech Enlisted Broadspire for Cheap Payouts
Exactech took deliberate steps to minimize payouts to patients harmed by their defective medical devices, a decision that significantly impacted the lives of those affected.
In an initial effort to avoid paying harmed patients what they truly deserved, Exactech chose early on to provide an upfront settlement option.
They hired Broadspire, a third-party settlement administrator and risk management company, to handle the claims reimbursement process. In doing so, Exactech sought to limit financial responsibility and reduce compensation amounts.
Patients were encouraged to file a claim with Broadspire to obtain reimbursement for their out-of-pocket medical expenses only.
Filing a claim through Broadspire would exclude critical compensations like lost wages, additional medical bills, and non-economic damages such as pain and suffering.
Exactech’s strategy was designed to pressure claimants into settlements that fell significantly short of what they truly deserved, a clear injustice.
Thus, a settlement through Broadspire would NOT compensate a victim for the total damages they suffered. Essentially, Broadspire was used to lure patients in by appealing to people’s desire for quick money without the need for a lawyer.
Exactech Faces Mounting Litigation Costs Over Harmful Implants
As time passed, Exactech’s legal troubles increased exponentially. Thousands of lawsuits were filed against the company by individuals who experienced life-changing adverse physical reactions from their defective implants.
According to Exactech, this generated high litigation expenses for the company. The added expenses jeopardized its cash flow and – among other factors – derailed the once profitable medical device business.
Facing escalating litigation costs that threatened its financial stability, Exactech took action to limit its liabilities.
Exactech Declares Chapter 11 Bankruptcy
On October 29, 2024, Exactech filed for Chapter 11 Bankruptcy in the U.S. Bankruptcy Court for the District of Delaware. This move is seen as an attempt to avoid liability to over 3,000 victims harmed by their defective orthopedic products.
The filing for bankruptcy has triggered an “automatic stay,” effectively halting all legal actions against Exactech, including cases in the MDL, Florida state court, and bankruptcy court.
This stay will remain in place as the bankruptcy court reviews Exactech’s financial situation and petition.
Additionally, the Broadspire program is no longer in effect, and all bellwether cases scheduled for trial will not proceed.
At this time, while court proceedings have been stayed, limited information is available.
Our Commitment to Holding Exactech Accountable for Defective Implants
Attorneys are awaiting critical decisions from the Bankruptcy Court regarding the company’s potential reorganization and the final deadline to submit proof of claim forms for each claimant.
The Yost Legal Group understands how concerning Exactech’s bankruptcy is for those affected. While this development impacts ongoing litigation, we remain committed to assisting our current Exactech clients in understanding their legal rights and next steps.
Our primary goal is to protect your best interests throughout this process.
Exactech Litigation Updates for Our Clients
If you are an existing client of The Yost Legal Group, we will continue to inform you about what is happening in the Exactech litigation.
You may have had or will require revision surgery. Please continue your medical care according to your doctor’s recommendations and keep us informed.
If you are a current client and have questions about the Exactech Defective Implant litigation, call us at 1-800-Yost-Law (1-800-967-8529).
We are unwavering in our commitment to holding Exactech accountable for their defective products. Our product liability attorneys will continue to post Exactech recall updates on the Exactech Recall Lawsuits.
Defective Medical Device Lawyers – Defective Product Lawyers – Product Liability Cases
Product Liability Lawyers – Product Liability Lawsuit – Product Liability Claims